ZAP QMS Software
Enterprise eQMS Software That Goes Beyond Record Keeping
Turn your quality management system into a connected operational part of the business — where documents, audits, risks, actions and compliance controls work together in real time.
Zebsoft provides a complete electronic quality management system (eQMS) software platform designed to support ISO 9001 compliance, FDA-regulated environments, GxP operations and enterprise quality management requirements across organisations of all sizes.
From document control and audits to CAPA, risk management and corrective actions, everything is managed within one connected quality management platform with full traceability and accountability.
Most QMS Platforms Store Information.
Zebsoft Helps Run the Operation.
Many organisations still manage quality through disconnected spreadsheets, isolated systems and reactive audit preparation.
Disconnected document control systems, isolated audit records, inconsistent training management and reactive corrective action processes create operational risk, duplication and reduced compliance visibility.
Documents exist. Procedures exist. Policies exist.
But proving that processes are actually being followed across teams, departments, suppliers and sites is far more difficult.
Zebsoft bridges the gap between quality documentation and operational reality. The platform helps organisations maintain audit readiness, manage operational compliance obligations and support continuous improvement activities across multiple departments, suppliers and locations.
Validation & Compliance Support
Validation documentation including IQ/OQ/PQ support, audit trails, electronic signatures and controlled workflow approvals are available for regulated deployments.
The platform can support computer system validation (CSV) approaches where required by operational or regulatory frameworks.
Operational Security & Data Integrity
Zebsoft includes role-based permissions, audit logging, controlled approvals and secure cloud hosting designed to support operational accountability and controlled information management.
AWS London hosting, encrypted communications and structured access controls help organisations maintain visibility and confidence in compliance-critical activities.
Built Once. Continuously Managed.
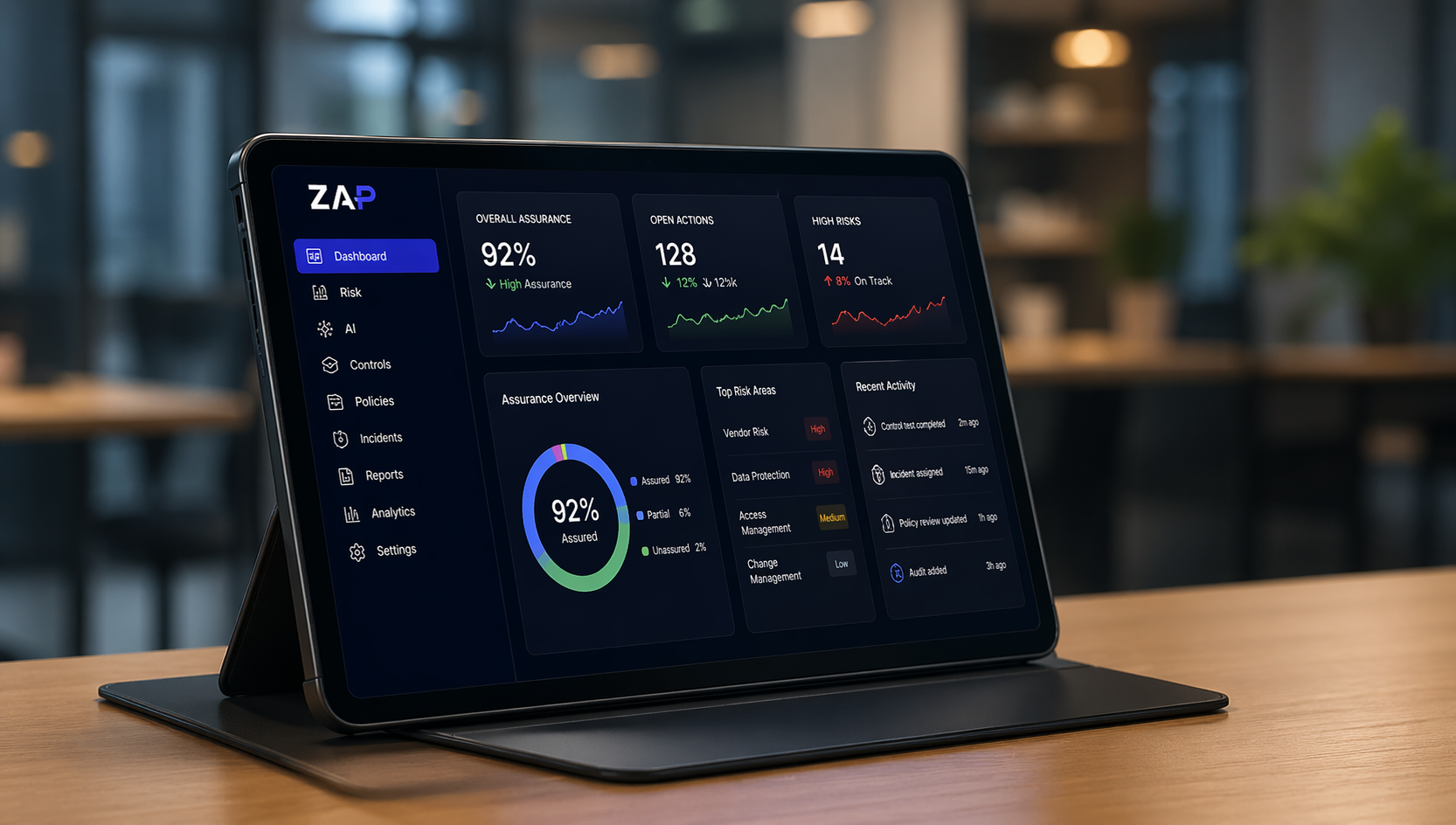
Monitor risks, audits, approvals, actions and compliance activity across the business in real time.

Live assurance, measured in real time.
92%
Assurance Score
+12% Over Last Month![]()
128
Open Actions
+27% Over Last Month ![]()
14
High Risks Identified
-7 Over Last Month![]()
85%
Control Testing Complete
+11% Over Last Month ![]()
Turn Your Quality Management System Into Operational Control
Book a live demonstration of the Zebsoft eQMS platform and explore how connected quality management can support compliance, accountability and operational performance across ISO 9001, FDA, GxP and enterprise quality management environments.


Audit Management

Risk Management

Document Control

Operational Oversight

Governance & Compliance

Continuous Validation
Your eQMS Should Do More Than Store Documents.
Zebsoft helps organisations maintain continuous visibility, accountability and operational control across quality-critical activities — supporting real-world compliance beyond static record keeping.
See how ZAP helps your business spot issues earlier, fix problems faster and stay ahead of risk.
Book a guided walkthrough of the Zebsoft Assurance Platform and see how audits, risk, controls and operational activity connect in one live system.














